 "We were able to dampen that effect and our severe cases are roughly 700 or less and have stayed stable, even though we still have days at 10,000 confirmed cases." 
Israeli health authorities expected severe cases to average 2,000 by late August, she said. 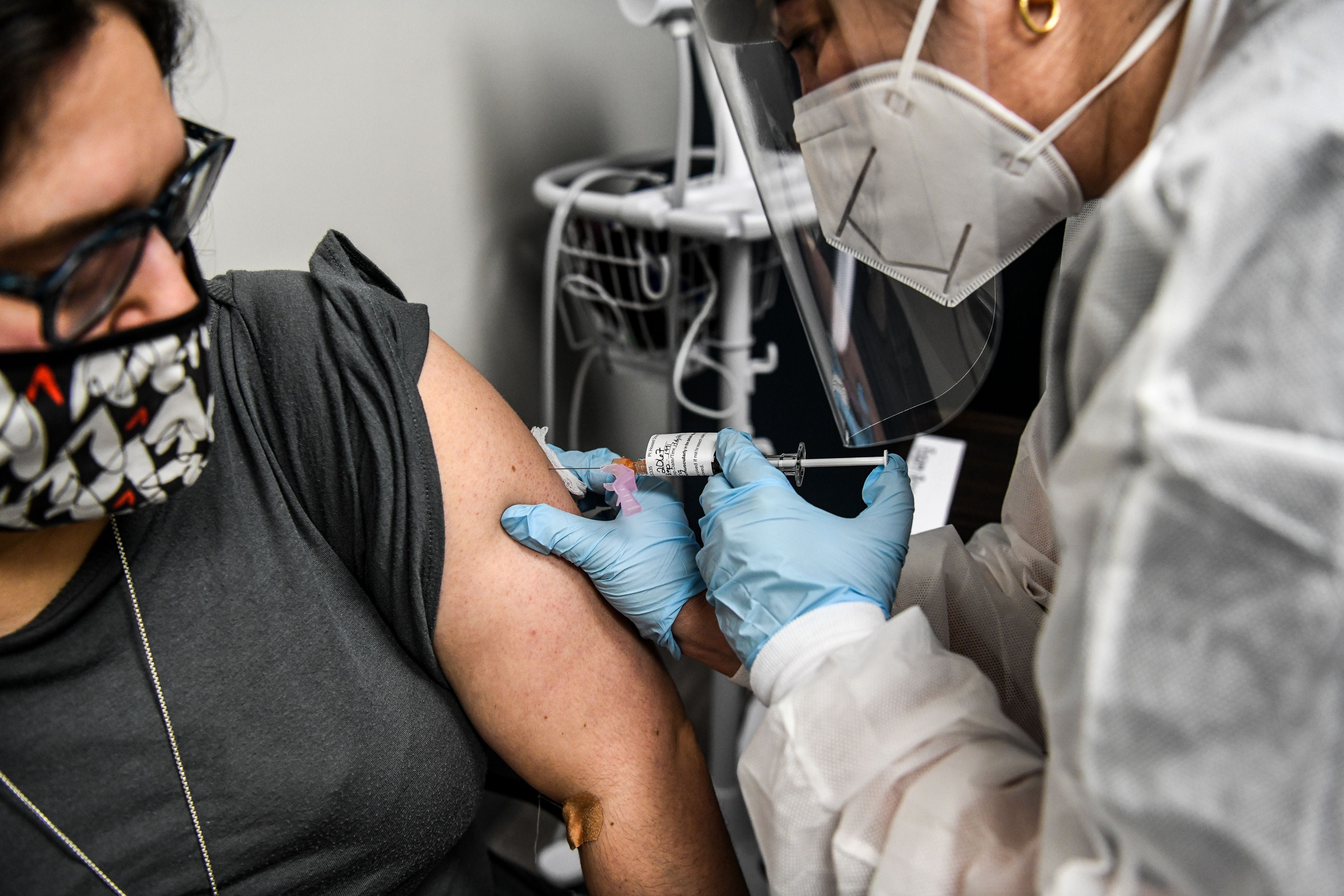
"We didn't want to wait to see those results and we knew that we needed to vaccinate a larger portion of the population in order to get the numbers down quickly," she told the committee. Health officials began to see a trend, she said, of individuals in their 40s and 50s who were fully vaccinated become critically ill with Covid. Sharon Elroy-Preiss of Israel's Health Ministry argued that if officials there had not begun distributing boosters at the end of July, the nation likely would have exceeded its hospital capacity. 30 when the fast-spreading delta variant was surging throughout the country. The data was collected from July 1 through Aug. In documents made public by the FDA on Wednesday, Pfizer said an observational study in Israel showed a third dose of the Covid vaccine six months after a second shot restores protection from infection to 95%. "That's part of the difficulty at looking at this kind of data without having the chance for FDA to review it," he said. He said the models used were complex and scientists have to ensure it "is giving you the correct results." Phil Krause, an FDA vaccine regulator and a co-author of The Lancet paper, was critical of the findings presented Friday, saying much of the data had not been reviewed by the federal agency or had not been peer-reviewed. Paul Offit, said he would support boosters for people over 60, but had trouble backing third shots for younger groups due to a higher risk of myocarditis.īefore the vote Friday, the committee listened to several presentations on data to support the wide distribution of booster shots, including from health authorities from Israel, where officials began inoculating the nation's population ahead of many other countries and began offering third shots to their citizens in late July. Hayley Gans, a voting member, said she was "struck" that the FDA was asking the committee to look at the totality of the evidence presented Friday because some data, including on safety, was still insufficient.Īnother member, Dr. Some members raised concerns about the risk of myocarditis in younger people, saying more research is needed.ĭr.  
In outlining plans last month to start distributing boosters as early as next week, Biden administration officials cited three CDC studies that showed the vaccines' protection against Covid diminished over several months. Senior health officials said at the time they worried protection against severe disease, hospitalization and death "could" diminish in the months ahead, especially among those who are at higher risk or were inoculated during the earlier phases of the vaccination rollout.īefore the vote, some committee members said they were concerned there wasn't enough data to make a recommendation, while others argued third shots should be limited to certain groups, such as people over age 60 who are known to be at higher risk of severe disease. The authors, including two high-ranking FDA officials and multiple scientists from the World Health Organization, argued Monday in the medical journal The Lancet that widely distributing booster shots to the general public is not appropriate at this time. In a paper published days before the advisory committee meeting, a leading group of scientists said available data showed vaccine protection against severe disease persists, even as the effectiveness against mild disease wanes over time. Personal Loans for 670 Credit Score or Lower Personal Loans for 580 Credit Score or Lower Best Debt Consolidation Loans for Bad Credit 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
